|
Altogether, the picture says that the 1s orbital on one atom and the 1s orbital on the other atom can combine in two different ways, producing the lower-energy, bonding σ 1sand the higher-energy, antibonding σ 1s*. The sides of the diagram just refer back to where those molecular orbitals came from, with dotted lines to guide you from one place to another. The order of energy so far is σ 1s, σ 1s*. It is analogous to the atomic orbital energy diagram (which goes 1s, 2s, 2p, 3s.). The middle of the diagram is just the molecular orbital energy diagram. What we see here is a molecular orbital interaction diagram. So, in a molecule, the lowest-energy molecular orbitals would be the ones formed from the lowest-energy atomic orbitals, the 1s orbitals. There are some departures from that rule, sometimes, but that's the simplest place to start. To a great extent, the order of molecular orbitals in energy can be considered to follow from the order of the atomic orbitals from which they are constructed. Just as we think of there being a progression of atomic orbitals from lowest energy to highest (1s, 2s, 2p, 3s.), we can organize these molecular orbitals by order of their energy. So far, we have looked at the ways in which pairs of atomic orbitals could combine to form molecular orbitals - to form bonds. it is not attracted by an externally applied magnetic field.\) On the other hand, the neutral #"C"_2# molecule has no unpaired electrons, so it is diamagnetic, i.e. it is attracted by an externally applied magnetic field. I won't show the calculation here because I'm not sure you're familiar with bond orders yetĪlso, an unpaired electron will make the #"C"_2^(-)# ion paramagnetic, i.e. Notice that because the extra electron is added to a bonding MO, the bond order of the #"C"_2^(-)# will actually be higher than the bond order of the neutral #"C"_2# molecule. The electron configuration of the #"C"_2^(-)# ion will be The electron configuration of the neutral #"C"_2# molecule is - I'll use the notation given to you in the diagram The lowest energy unoccupied molecular orbital is #2p_(sigma)#, so that is where the extra electron will be added. It will be added to the lowest energy unoccupied molecular orbital, or lowest unoccupied molecular orbital, #"LUMO"#, that follows that highest energy occupied molecular orbital, or highest occupied molecular orbital, #"HOMO"#.Īs you can see in the diagram, the two #2p_(pi)# orbitals, let's say #2p_(pix)# and #2p_(piy)#, are the highest energy occupied molecular orbitals. So, where would this extra electron go in terms of molecular orbitals? It thus follows that the #"C"_2^(-)# species will have This, of course, implies that a #"C"_2# molecule has a total of  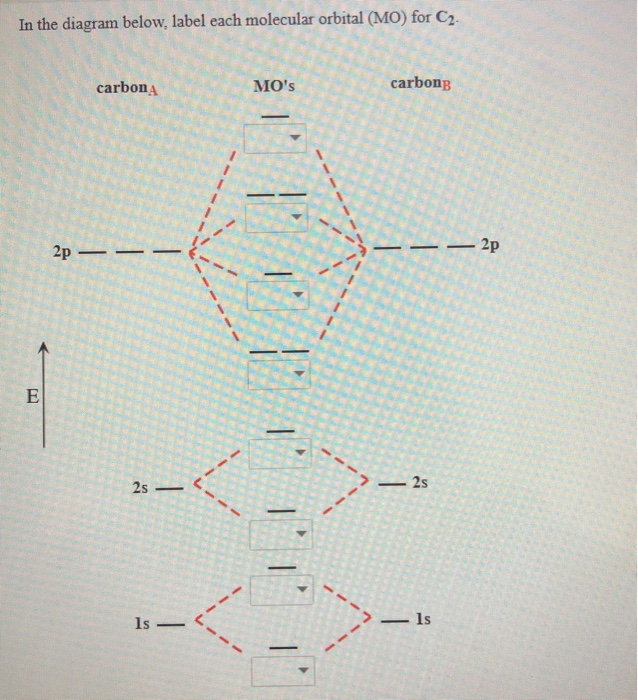
As you know, a neutral carbon atom has a total of #6# electrons. You need to add an electron and not remove one because of the overall negative charge that exists on the molecule. The problem provides you with the MO diagram for the #"C"_2# molecule, so all you really have to do here is add an electron to that diagram.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
